Algal symbiosis and mass bleaching
Algal symbiosis and mass bleaching

Algal symbiosis
Symbiosis, the interdependence of different organisms for the benefit of one or both participants, is prevalent in the oceans as it is on land. Within the spectrum of marine symbioses, zooxanthellae clearly have a special, and exceptionally long-standing, relationship with numerous partners.
Scleractinian corals and zooxanthellae originated and diversified together, between 140–200 mya. Zooxanthellae are not just found in Scleractinia, however, and also occur in other cnidarians (soft corals, anemones and their allies) as well as in an assortment of other animals encompassing single-celled ciliated protists, sponges, flatworms and molluscs (including giant clams). Once thought to be a single dinoflagellate species, Symbiodinium microadriaticum, zooxanthellae have been found to be genetically diverse (consisting of many genetic types or 'clades'), and new Symbiodinium species are described regularly, although they all look similar.
Detailed morphological and genetic analyses have recently revealed a wealth of different zooxanthellae species, and the divergent Symbiodinium clades have recently been partitioned into multiple genera, including Breviolum, Cladocopium, Durusdinium and Gerakladium, better reflecting their long evolutionary history (LaJeunesse, Parkinson, Gabrielson et al., 2018).
They can all live independently, although not in such concentrated numbers nor with such long-term security as they can in the tissues of hosts. In corals (but not clams) they live inside the cells of the host organisms — in the innermost (gastrodermal) layer of the two cell layer body wall. All zooxanthellae are tiny, (approximately one hundredth of a millimetre in diameter), and only one to four occur within each coral cell.
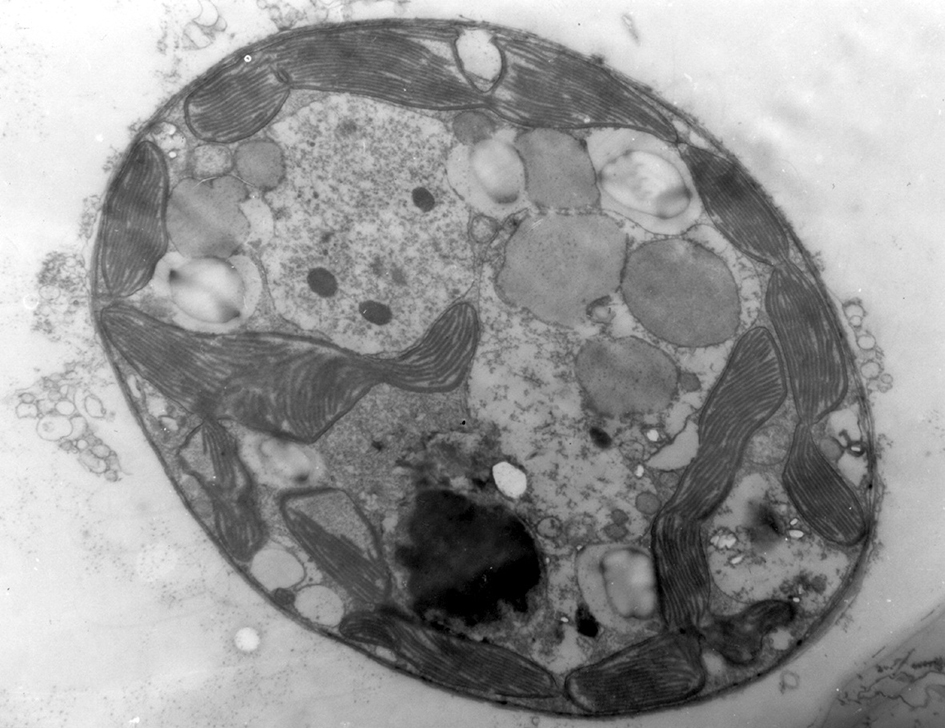
An electron microscope image of a zooxanthella. Photogragh: Ove Hoegh-Guldberg. Figure 1.
Zooxanthellae photosynthesise as do other plants, with up to 95% of the nutrients they produce being leaked directly to the host organism. Nevertheless corals are voracious feeders, capturing zooplankton using the stinging batteries of nematocysts housed on their tentacles. Most species feed only at night, when the zooxanthellae are not photosynthesizing. Therefore corals have two very different food sources, termed autotrophy and heterotrophy respectively. The contributions to nutrition from each source vary in relation to ambient conditions. However, many if not most corals that are kept in darkness (so that their zooxanthellae cannot photosynthesise) will start to die after a few months irrespective of how much food they have from elsewhere.
The main points about coral - algal symbiosis are (and see, for example, the general reference van Oppen and Lough, 2018):
- Corals acquire their zooxanthellae either directly from the parent colony or through infection into free-swimming or newly-settled larvae, termed 'vertical' and 'horizontal' transmission respectively. Vertical transmission is most commonly found in coral species that brood larvae, whereas the larvae of broadcast spawning species tend to acquire zooxanthellae 'horizontally' from the water column.
- In some species uptake of non-parental zooxanthellae in early life may be by random chance, giving different advantages to different colonies: some colonies might be infected with temperature tolerant symbionts, others with more productive ones. In other species, corals preferentially select particular zooxanthellae types form those available in the water column. The uptake of different zooxanthellae types is an important recent discovery, as it was initially believed that all corals hosted the same zooxanthellae, a logical conclusion when just one zooxanthellae species was described (see Gong, Chai, Xiao et al., 2018 and Stanley and van de Schootbrugge 2018).
- Zooxanthellae readily change in abundance depending on conditions such as season, position on the coral and light level (as seen in corals experimentally transplanted to different water depths).This is consistent with corals having the capacity to regulate the numbers of zooxanthellae in their tissues, the mechanisms of which remain to be fully explained.
- More than one genetic type of zooxanthellae can occupy a single colony. Many, perhaps most, coral species can host several different zooxanthellae types in their tissues.
- The abundance of genetic types varies geographically on any scale.
- Some genetic types facilitate a faster coral growth rate than others.
- Different types vary in their metabolic responses to different kinds of stress, including abnormally high or low sea temperatures, levels of illumination or salinity, resulting in different levels of oxygen production and toxicity, and the 'bleaching' response of the host coral. Hence, the specific and sub-specific identities of resident zooxanthellae can influence their host’s susceptibility and thereby contribute to broad disparities in thermal tolerance among individual coral colonies and species.
- Some corals can 'shuffle' or 'switch' their zooxanthellae populations in response to environmental change. Shuffling refers to changes in the proportions of different zooxanthellae types in a coral's tissues. Switching refers to the replacement of zooxanthellae, typically following bleaching, with a different type acquired from the water column. These are important areas of current research, offering a small kernel of hope in terms of the ability of corals to acclimate and/or adapt to the increasing environmental impacts of the 21st century, and advances in understanding are happening rapidly.
- However, other studies showing that the genetic history of corals and that of zooxanthellae closely match, argue against any medium-term history of adaptation by the changing of symbionts.

Bleaching
Coral tissue that has turned white or pale is described as 'bleached'. This phenomenon has a very broad spectrum of effects in its appearance and impact on corals. Minor bleaching can occur in small patches on a few individual colonies on otherwise unaffected reefs. Moderate bleaching can occur on some or all parts of some colonies of the more susceptible coral species while others remain unaffected. Major, or mass, bleaching can affect virtually all corals on whole tracts of reefs, particularly their shallower slopes. From the air, entire reef tracts can appear a ghostly white colour, as corals expel their zooxanthellae en masse. During this phase, corals can initially appear 'fluorescent', as they pale progressively to stark white. Within weeks, corals that have died are coated with algae, and the reef becomes a graveyard of dull brown coral skeletons trailing strands of algae waving in the current.
Bleaching is a symptom of stress in the coral and/or the coral's zooxanthellae. Much of the natural patchy bleaching occasionally seen in otherwise healthy corals is unimportant as it causes little or no long-term problems for a colony. Such patches may be due to a temporary loss of photosynthetic pigment from the zooxanthellae, predators of many sorts, or attacks from neighbouring colonies in the battle for growing space. Colony-wide bleaching is clearly of more concern as this can result in the death of the coral. Such bleaching has a number of causes including disease, lowered salinity, high temperature or increased light (usually as a result of very calm sea conditions and/or turbidity decrease). In each case, severe bleaching indicates that the coral's tissue integrity has been compromised. Although any of these causes can result in widespread mortality, by far the most common and most conspicuous cause is a combination of high light and elevated temperature. Where this occurs across whole reef tracts it is known as 'mass bleaching' which often leads to mass death of corals.
Such temperature/light-induced bleaching usually begins with excessively elevated levels of photosynthesis which leads zooxanthellae to produce toxic levels of oxygen. This occurs most commonly in shallow reef waters where corals are exposed to a combination of high sea-water temperatures and high levels of sunlight, for corals can regulate the quantity of zooxanthellae in their tissues, but not the rate at which these produce oxygen. Under conditions of high temperature and excess light, zooxanthellae become poisonous, actually deactivating the sunscreens corals produce to protect themselves. After studying this process in detail, coral physiologists have discovered it to be a general phenomenon also found in terrestrial plants as a mechanism for protecting leaf tissue from excess oxygen through over-exposure to sunlight.
In corals exposed to extreme levels of solar radiation in general and ultra-violet light in particular, together with high water temperatures (usually associated with shallow ponded water), zooxanthellae produce oxygen 4-8 times faster than the coral host can take it up. When this happens, some oxygen ceases to play a normal role in photosynthesis and becomes chemically active as oxygen radicals causing cellular distress. As oxygen radicals become toxic, the zooxanthellae that produce them are expelled by the corals even though this action too puts the corals at risk. Expulsion most commonly takes place by sloughing-off the gastrodermal cells in which the algae live, but there are also other mechanisms like resorption and tissue death. Such discoveries explain why bleaching is light - as well as temperature-dependent. They also explain a host of experimental variations correlated with environment, colony characteristics (shape and species) and the place and depth where experimental corals were originally collected. Additionally they help to explain why some corals die after they bleach: even when other nutrients are present and there is plenty of food in the form of zooplankton they may simply not have enough gastrodermal cells to function.
The temperature/light conditions which cause corals to expel their zooxanthellae are normally not localised and commonly affect a significant portion of the reef experiencing the temperature pulse. This can result in widespread mass bleaching of coral colonies, particularly at shallow depth.

Mass bleaching
Mass bleaching was first recorded in early 1980 in the Caribbean and surrounding seas (notably in Jamaica and the Bahamas), the far eastern Pacific (Panama and the Galápagos Islands) and in isolated instances in the Pacific (notably French Polynesia and Thailand), and on the Great Barrier Reef.
There have been seven major bleaching events on the Great Barrier Reef, the three spanning the summer seasons of 1981/82, 1997/98 and 2001/02 being the most widespread, at least prior to the recent 2016 and 2017 years. However, it was the 1981/82 mass bleaching event that really drew attention to the association between ocean temperature and bleaching.

A community of bleached coral, mainly Montipora (left) and Acropora (right). Photograph: Katharina Fabricius. Figure 2.
The 1997/98 mass bleaching event was extraordinarily widespread. It affected reefs in over 50 countries throughout the Pacific and Indian Oceans, the Red Sea and the Caribbean, and even affected corals in high latitudes including those of the Red Sea and Arabian Gulf.
On the Great Barrier Reef, this mass bleaching coincided with the highest sea surface temperatures ever recorded. It was also at this time that 500-year-old Porites colonies died, strong evidence that mass bleaching had human causes.

A bleached Porites colony several hundred years old. Photograph: Katharina Fabricius. Figure 3.
The third major mass bleaching event took place in 2001/02, again affecting many countries. By this time it had become clear that there was a causal link between the periodicity of global bleaching and changes in ocean temperature associated with El Niño Southern Oscillation cycles. The gap between these events appears to be closing as the difference between El Niño years and non-El Niño years diminishes, a process that has started occurring and is destined to continue on into this century. Importantly, while 2006 was not an El Niño year, there was significant mass bleaching of corals on the Great Barrier Reef. Extensive mass bleaching occurred in several countries of the Caribbean in both 2005 and 2006. Present indications are that increases in global temperatures will lead to mass bleaching in all years by 2030. The latest global event which occurred from 2015-17 was the most severe on record, and does nothing to assuage these concerns.

Mass bleaching and the environment
Some of the important features of mass bleaching are as follows.
Initiation
- Because mass bleaching is caused by the inability of corals to process all the oxygen from light produced by photosynthesis, it occurs in corals exposed to strong sunlight.
- Corals become susceptible to bleaching if the water temperature exceeds or drops below historical limits. What matters is the amount and duration of heating or cooling at and beyond these limits rather than absolute temperature as this varies geographically.
- Short periods of high temperature may be more stressful than longer periods of a slightly lower temperature but bleaching stress is basically cumulative over periods of weeks to months rather than a sudden all-or-nothing reaction.
- Bleaching may be caused by stresses other than temperature. These include increased ultra-violet radiation as distinct from solar radiation, lack of water movement around the colonies, low salinity after floods, bacterial infection, sudden chilling, and chemical pollutants. For example, mass bleaching on nearshore reefs of the Great Barrier Reef has been triggered by rapid declines in sea temperature and salinity, when a tropical storm traversed the reef tract, producing cool freshwater flood runoff from the adjacent mainland. However, given Earth's current trend of global warming, it is the combination of high temperature and high solar radiation (particularly its ultra-violet component) that is causing maximum bleaching at the scale of ocean basins, unprecedented, at least in the historical record.

This bleached Goniopora may recover. The white bleached polyps are already smaller than the pale brown polyps which still have some zooxanthellae. Photograph: Katharina Fabricius. Figure 4.
Geographic variation
- Corals growing on equatorial reefs are more tolerant of heat than those growing at higher latitudes. For example, corals bleach at lower temperatures on the southern Great Barrier Reef than they do in the north, indicating the importance of temperature thresholds.
- Bleaching can occur in high latitude coral reefs including Hawaii, Bermuda and Lord Howe Island.
- There is local geographic variation in the extent of bleaching within the same species.
- Susceptibility to bleaching is affected to a significant extent by the history (experience) of individual coral colonies.
Taxa affected
- Some groups of corals are more susceptible than others. The families Acroporidae and Pocilloporidae are commonly the most affected as is the hydrocoral Millepora. The least affected are the corals with massive growth forms, notably the faviids. There are, however, many reported differences in detail among studies. There have been several imaginative ideas about this, one being that least affected species belong to the oldest families, another that the faster growing corals are most susceptible, another that it is mostly a matter of tissue thickness and associated densities of zooxanthellae. However, all corals with zooxanthellae will bleach if conditions become severe enough for long enough.
- Mass bleaching is most common and most severe in shallow communities but it is not restricted to the shallows. Major occurrences have been recorded to depths of over 25 metres on the Great Barrier Reef and 30 metres on Scott Reef off Western Australia.
- Mass bleaching on the Great Barrier Reef is more common in inshore communities, but even within the same region it is occasionally more severe offshore. It may be particularly prevalent in intertidal pools, although there are notable exceptions.
- Corals that are aerially exposed at low tides are the most heat (and light) tolerant of all and often do not bleach when those in nearby deeper water do.
Physiological effects
- Bleaching inhibits coral growth, but this is temporary if the stresses are removed and normal levels of zooxanthellae return.
- Bleaching can disrupt the reproductive cycle, an outcome which has the potential to inhibit recovery of some coral species and thus change their relative abundance.
- Lower energy reserves caused by repeated bleaching may have contributed to disease outbreaks that have greatly affected the corals of Florida and the Caribbean. This is now being seen increasingly on the Great Barrier Reef.
- For healthy reefs, there are many accounts of recovery of bleached corals and this may occur in as short a time as several months. Even completely white corals sometimes recover. This has been attributed to the survival of very low numbers of zooxanthellae in basal parts of coral colonies, which subsequently repopulate the coral's tissues. Another possibility is that surviving corals obtain a new suite of zooxanthellae via horizontal transmission from the water column, having expelled their prior complement of zooxanthellae during the bleaching episode.
- Recovery of coral communities in which most corals have died, measured by restoration of coral cover and species composition, normally takes a decade or more in pristine environments, depending on larval supply, algal take-over after bleaching, and other local factors. Restoration of coral population age structures may take much longer, from decades to centuries, on reefs where the oldest corals, such as centuries' old massive corals, were killed by bleaching.
- Some genetic types of zooxanthellae, mostly members of the genus Durusdinium (formerly Clade D), appear to be more resistant to high levels of light and/or temperature (up to 1-1.5C) than others although this also remains controversial.
- Mass bleaching and ocean acidification are likely to have future synergistic effects.
Links to El Niño events
There is no clear link between enhanced greenhouse warming and the frequency or intensity of El Niño events. Analyses of historical records and projections from General Circulation Models are both ambivalent on the subject. The general climatic changes accompanying El Niño development are fairly well understood, although the factors controlling their initiation, intensity and periodicity remain obscure. The 1997-98 El Niño event was the most extreme in recorded history yet it is still possible that this and the two other major events in the past two decades (1981-82 and 2001-02) were exacerbated by other, slower, climatic cycles which are part of the natural variability of the Earth's climate and not a response to greenhouse warming. The recent global events of 2016 and 2017, however, have clear causal links to our warming planet, above and beyond that of the underlying El Niño cycles.
Although any direct causal link between enhanced greenhouse warming and El Niño intensity and frequency is uncertain, it is clear beyond doubt that El Niño cycles and mass bleaching are connected. Mass bleaching is not caused by a direct overall increase in ocean temperature but by short term concentrations of heat in the affected areas. On the Great Barrier Reef these temperature increases can be caused by El Niño events which pulse oceanic water from the Western Pacific Warm Pool, perhaps 1-2°C above what was once normal, into coastal regions. If this water is then trapped in the lagoon of the Great Barrier Reef it can warm still further, exacerbating the effects of the original pulse.
Essential points about El Niño, global ocean temperatures, and mass bleaching are:
- Enhanced (anthropogenic) greenhouse warming is occurring as a result of the amount and rate of build-up of CO2.
- Despite the oceans absorbing most of the heat generated by global warming to date, they are warming far more slowly than the atmosphere because of thermal inertia. A significant proportion of this warm water is remaining in the surface layers of tropical oceans because it is being accumulated too rapidly to be adequately dispersed and mixed throughout the ocean depths by natural water circulation.
- Thermal energy from enhanced greenhouse warming of the oceans is concentrated into the Western Pacific Warm Pool, the world's largest mobile body of heat.
- El Niño events themselves are not the result of global warming; they are a natural phenomenon. However, there is continuing debate over the role of enhanced greenhouse warming in altering the frequency and/or intensity of El Niño events.
- The association between El Niño events and mass bleaching will be a temporary one as far as most reefs are concerned. As the Western Pacific Warm Pool widens and deepens, its central core will increasingly affect more extensive outer reef regions in non-El Niño years. At this point the thermal peaks currently delivered by El Niño will become less and less exceptional until they are irrelevant. This process is already beginning for the Great Barrier Reef and similar observations have been made in other parts of the world.
- Although El Niño oscillations will become irrelevant for the Great Barrier Reef, they may continue to play a role in transmitting warm water pulses to other regions of the world for some time to come as the Warm Pool deepens and widens.
- We have already entered the time-frame where mass bleaching is occurring without El Niño enhancement. If the relationship of atmospheric CO2 levels to the amount and/or frequency of bleaching continues on its present trajectory, there will be a point where every year will have a similar impact on corals as the worst El Niño events have had in the past.
Acclimatisation and adaptation
Acclimatisation (where the individual's tolerance of environmental conditions increases during their lifetime) and adaptation (an evolutionary process involving natural selection through differential reproduction and survival) are seemingly the only escape routes corals have from the warm world of the future. As introduced above, the recent discoveries of multiple types of zooxanthellae, and corals' inherent capacities to shuffle and/or switch zooxanthellae, may provide additional avenues for both acclimation and adaptation to increasing climate disruption, ocean acidification and other impacts this century.
Acclimatisation There is evidence on both local and global scales that the same and/or closely related coral species show different tolerances to temperature in different locations. On local scales, good examples are corals that tolerate the very high temperatures found in intertidal pools, in water around natural thermal vents or close to thermal outlets of power stations. Normal maximum water temperatures found in particular geographic areas play a large role in determining tolerance to bleaching. Whole suites of corals can survive short-term peaks of 36°C in the Arabian Gulf, parts of the southern Red Sea and sporadically elsewhere. Like most animals, corals may acclimate to tolerate these temperatures by altering biochemical pathways. On local scales this process is likely to be due to acclimatisation whereas across widely separated geographic areas there may be a larger component of genetic selection, especially where local tolerance to extreme conditions is involved.
Adaptation Corals were probably once adapted to higher temperatures in the geological past. However the template of todays' oceans is so different from those of the remote past that most meaningful comparisons are questionable. It is nevertheless worth revisiting the fossil record at this point, and particularly the major 'reef gaps' that extended for millions of years following the mass extinctions. The Scleractinia first rose to prominence after one such extinction, the end-Permian, and have survived those of the Mesozoic, albeit with significant changes in lineages via reticulate evolution and natural selection, and major reductions in reef-building during periods of major climatic instability. Today, the largest uncertainties relate to future rates of global warming and ocean acidification. If our collective global emissions of fossil fuels and other sources of greenhouse gases continue, along with positive climate feedbacks including loss of polar albedo, melting of permafrost and release of 'ice gas' methane clathrates off continental shelves, the climate system will likely be driven ever more rapidly beyond a tipping point. Under this scenario, it is unlikely that corals will have time to adapt. Levels of future extinctions may be significant, and reefs will cease to grow - effectively forming another 'reef gap' in the fossil record of the distant future.


